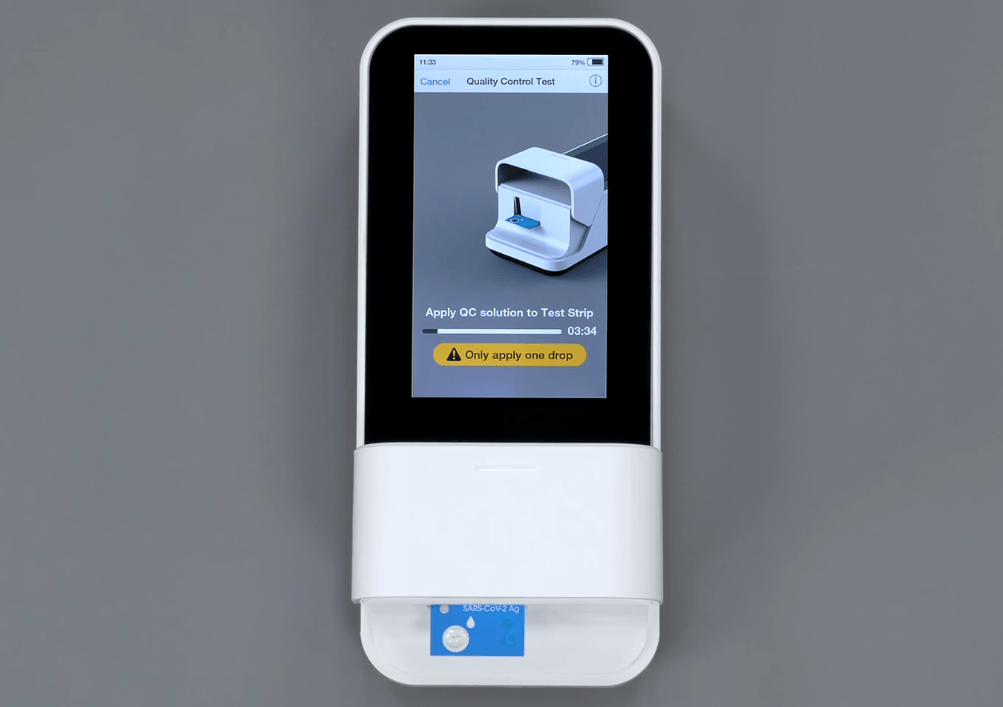
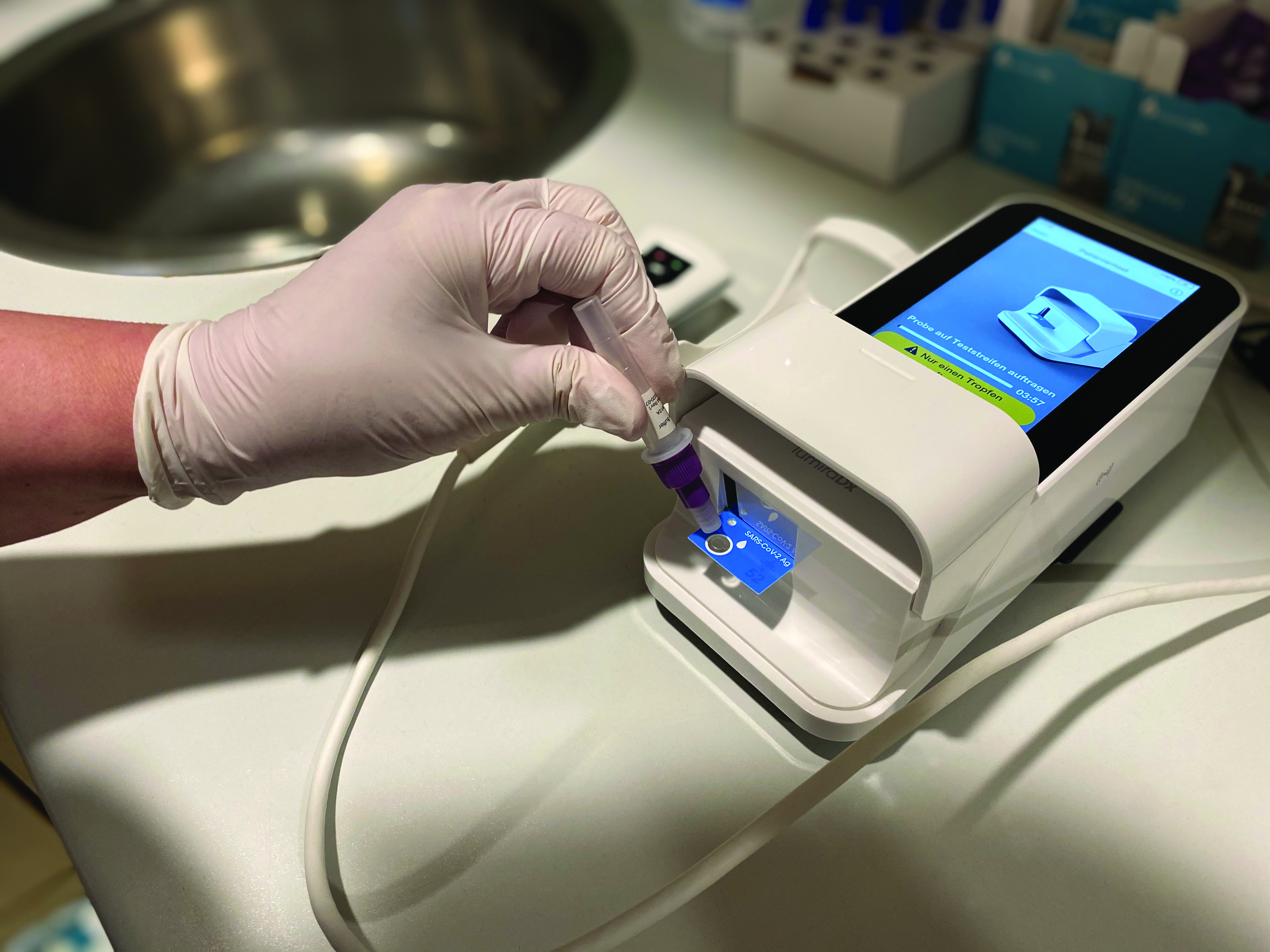
A rapid, high-sensitivity SARS-CoV-2 nucleocapsid immunoassay to aid diagnosis of acute COVID-19 at the point of care | medRxiv
LumiraDx launches C-Reactive Protein test to combat antimicrobial resistance in India, ET HealthWorld
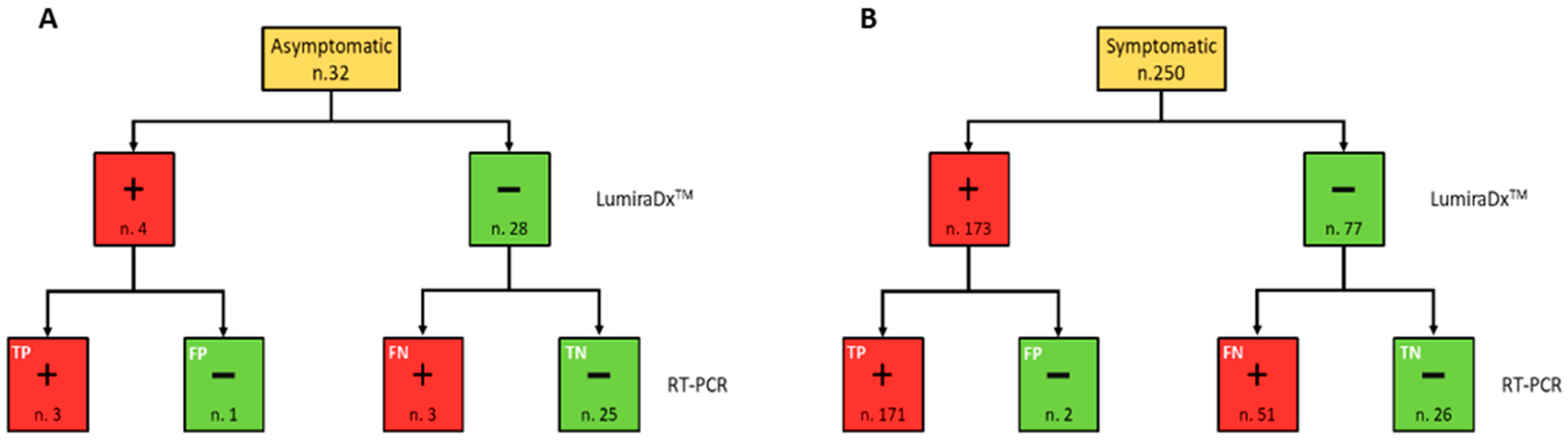
IJERPH | Free Full-Text | Rapid Antigen Test LumiraDxTM vs. Real Time Polymerase Chain Reaction for the Diagnosis of SARS-CoV-2 Infection: A Retrospective Cohort Study

LumiraDx Expands its Cardiovascular Testing Portfolio with CE Marking of its NT-proBNP Test and new Exclusion Claim for its D-Dimer Test

LumiraDx riceve l'autorizzazione per il test antigenico SARS-CoV-2 in Giappone e Brasile; l'Italia raccomanda l'espansione del test antigenico basato su microfluidica